Diabetes is a chronic disease related to blood sugar levels. When you have diabetes, you must constantly monitor your blood sugar and use insulin. Technologies like the Omnipod system can facilitate this management and improve care for people with diabetes.
What are Omnipods?
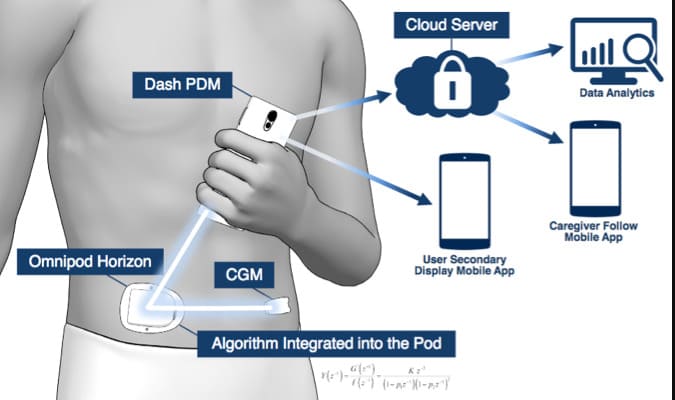
The Omnipod is a tubeless, waterproof, and wearable insulin pump manufactured by Massachusetts-based Insulet Corporation.
It is an alternative to traditional insulin pumps and an effective insulin treatment. The company recommends the Omnipod uses its proprietary Pod Therapy technology to deliver insulin continuously for about 3 days without the need for multiple injections.
Teams can deliver rate-based doses of rapid-acting insulin that a person programmed into a Personal Diabetes Management (PDM) device. People can also combine Omnipod technology with a continuous glucose monitor (CGM) to create an automated insulin delivery (AID) system.
An AID system, which some may call an artificial pancreas or a hybrid closed-loop system, can help mimic the function of a healthy pancreas. Research shows that automated insulin delivery systems can significantly improve diabetes management in adults, children, and adolescents.
Omnipod helps patients save more money
The Omnipod provides a continuous delivery of insulin through a waterproof insulin pump called a Pod – all without the need for multiple daily injections. Get insulin continuously and freely for 3 days (up to 72 hours) with these innovative features:
- Wearable: Place the Pod almost anywhere you normally inject
- Water proof*: Take your insulin wherever life takes you, even while swimming
- Anti-tangle: No need for tubes of traditional insulin pumps
The Omnipod 5 AID system is still awaiting approval from the Food and Drug Administration (FDA), but early researchTrusted Source shows it can significantly improve blood glucose measures in people with diabetes. diabetes.
The results of a 2021 multicenter clinical study funded by Insulet show that the Omnipod 5 system can significantly improve diabetes outcomes in children and adults. After three months of using the system, the participants had:
- Lower rate of hypoglycemia
- Lower rate of hyperglycemia
- Increase time in range, or time spent in target blood sugar range
- Improves HbA1c levels, which measure average blood sugar over the previous 2–3 months
Research suggests that the device may also help improve quality of life. According to diaTribe, 92% of adults and adolescents and 99% of children chose to continue using the Omnipod 5 after completing the trial.
Another 2021 outpatient review funded by Insulet also found that the Omnipod 5 system is safe and works well in people 6–70 years of age at all target glucose levels. Additionally, a 2021 paper highlights the safety of the Omnipod 5 system in young children with type 1 diabetes. It’s important to note, however, that Insulet funded the study and some of the authors. related to the company.